
Phosphorylation of UVRAG at Ser498 by mTORC1 enhances the UVRAG–Rubicon interaction, where Rubicon acts as an antagonist of UVRAG/HOPS-mediated autophagosome and endosome maturation 15. UVRAG stimulates lysosomal fusion of autophagosomes and endosomes by binding to the homotypic fusion and vacuole protein sorting (HOPS) complex 5. In addition, Rubicon interacts with UVRAG to inhibit lysosomal fusion with autophagosomes and endosomes. Rubicon directly binds to the PI3K-UVRAG complex through its RUN domain and contributes to the suppression of autophagy initiation by inhibiting PI3K activity 14. Rubicon regulates both the initiation of autophagy and autophagosome maturation 2. Rubicon was first identified as a Beclin 1-interacting protein. UVRAG plays a role in endosomal flux of cargo into lysosomes and autophagosome maturation by assembling class C type VPSs without Beclin 1/VPS34 13. It is unclear whether UVRAG-associated PI3K can control the maturation of autophagosomes. The PI3K-Atg14L complex is localized at the ER, endosome, and isolation membrane and is essential for autophagosome formation, whereas the PI3K-UVRAG complex is localized primarily in the late endosome, thereby mediating endosomal trafficking 12. The PI3K complex also contains either Atg14L or UV radiation resistance-associated gene (UVRAG). The PI3K complex is composed of vascular protein sorting (VPS) 34, a catalytic subunit, Beclin 1, a scaffold protein, and VPS15, along with several additional interactors to initiate autophagosome nucleation. Activated Ulk1/Atg1 directly phosphorylates AMBRA1, a Beclin 1-interacting protein, inducing translocation of the class III phosphatidylinositol 3 kinase (PI3K) complex to the endoplasmic reticulum (ER), where it initiates autophagosome formation 11. This inhibition is followed by the activation of the Unc-51-like kinase 1 (Ulk1)/Atg1 complex, consisting of Ulk1, Atg13, FIP200, and Atg101 10. Autophagy is initiated by the activation of an energy sensor, AMP-activated kinase (AMPK), which in turn inhibits mammalian target of rapamycin complex 1 (mTORC1) expression. Cellular components are engulfed into a double-membrane structure, the autophagosome, in either a nonselective or selective manner, and the cargos are digested after fusion of autophagosomes with lysosomes 9. Rubicon is involved in multiple cellular functions, including autophagy, endosomal trafficking, phagocytosis, and inflammatory responses.Īutophagy is a highly conserved catabolic process in eukaryotes orchestrated by more than 35 Atg proteins that were first identified in yeast 3. We also discuss the possible role of Rubicon as a new therapeutic target for various cardiac diseases.įull size table Rubicon in canonical autophagy Here, we review the current understanding of Rubicon function in canonical and noncanonical autophagy and summarize the roles of Rubicon in the heart. Recent studies have identified the involvement of Rubicon in heart diseases 7, 8. Rubicon may be involved in disease progression through its effects on canonical or noncanonical autophagy (Table 1). On the other hand, Rubicon positively regulates LC3-associated phagocytosis and endocytosis, noncanonical forms of autophagy, in response to extracellular molecule stimuli 4, 6. Rubicon inhibits autophagy and endosomal trafficking by directly interacting with Rab7 5. In 2009, two groups identified Rubicon as a protein that suppresses autophagy by interacting with the Beclin 1 complex 2, 4. 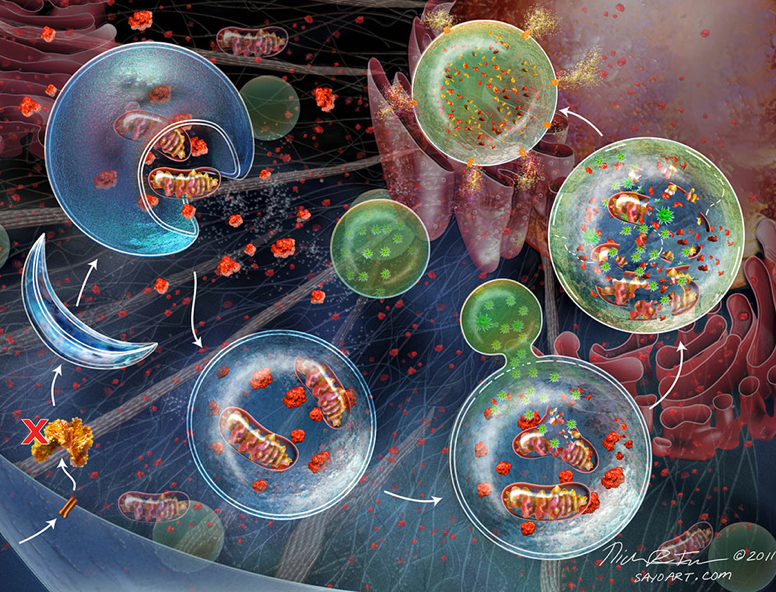
Although most Atg proteins positively regulate autophagy, together with Bcl-2, another autophagy regulator, Rubicon is one of the few known endogenous negative regulators of autophagy 2, 3. Autophagy is tightly regulated by more than 35 kinds of au topha gy-related (Atg) proteins. Therefore, maintaining autophagy function is an important goal for the development of therapeutics. Failed autophagy has been linked to aging, cancer, metabolic disorders, and heart disease 1. Autophagy is modulated during cellular stress to delay the onset of various diseases, including heart diseases. Degradation of cellular components and organelles through autophagy helps to maintain cellular homeostasis.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
